Injectable (mainly subcutaneous, intramuscular and intravenous routes, may include other routes if applicable e.g. epidural, intra-articular, intravitreal, intrathecal and intramyometrial)
Intravenous infusion of a defined dose (volume), over a specified infusion duration.
Approved by PSWG (Parenteral Services Working Group) Jan 19, 2017
Intravenous medication of a defined concentration, infused at a specified rate; variable duration is determined by clinical endpoint(s).
Approved by PSWG Jan 19, 2017
IV drug infusion for which the administration rate (dose) is adjusted to achieve or maintain a desired clinical response for an unspecified duration.
Approved by PSWG Jan 19, 2017
A defined dosage of IV medication or volume of fluid, infused over a specified duration (or rate), as a loading dose, single treatment or at any time during a continuous IV infusion of the same medication to achieve rapid clinical response.
Approved by PSWG Jan 19, 2017
Manually controlled administration of medication from a syringe connected to an IV access device; this may include IV doses manually administered in an emergency.
Approved by PSWG Jan 19, 2017
A method of preparing a sterile parenteral product, starting with an empty IV bag (or other container) to which a precise (drug) volume and diluent volume is added. There is no overfill with full sterile compounding.
Approved by PSWG Dec 10, 2015
(Applies to preparation of continuous IV infusions, other than full sterile compounding)
When the additive (drug) volume is greater than 10% the diluent volume (reported on the container), a diluent volume equal to the drug volume (or otherwise stated in the treatment protocol) is removed from the container prior to adding the drug.
Revised and approved by PSWG May 12, 2017
Applies to preparation of intermittent infusions; entire contents of the container are administered to ensure patient receives full dose
Approved by PSWG Dec 10, 2015
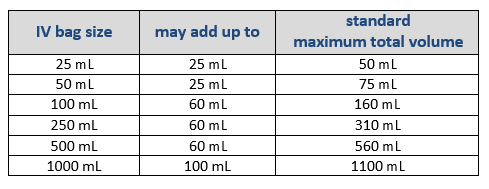
Volume added to Commercial IV Bags
- Associate Lead, Medication Management, Clinical & Systems Transformation (Elfreda Chan); regarding standardization requirements for building Cerner IV order set.
- Alaris System Direction for Use – with v9 Model 8015 – Programming (July 2010)
- Parenteral Drug Therapy Manuals – Vancouver Coastal (Jan 2008), Richmond (Sept 2007), Vancouver Acute (June 2009), Providence Health Care (Dec 2008)
- BC Women’s Parenteral Therapy Manual
- Fraser Health Parenteral Drug Administration Guidelines (Nov 2011)
- Proceedings from the ISMP Summit on the Use of Smart Infusion Pumps (2009)
- ISMP Addressing a Trefecta of Overlooked IV Medication Risks (2015)
- ISMP Safe Practice Guidelines for Adult IV Push Medications (2015)
refer to Elsevier Clinical Skills
Last Revised: 9 March 2023